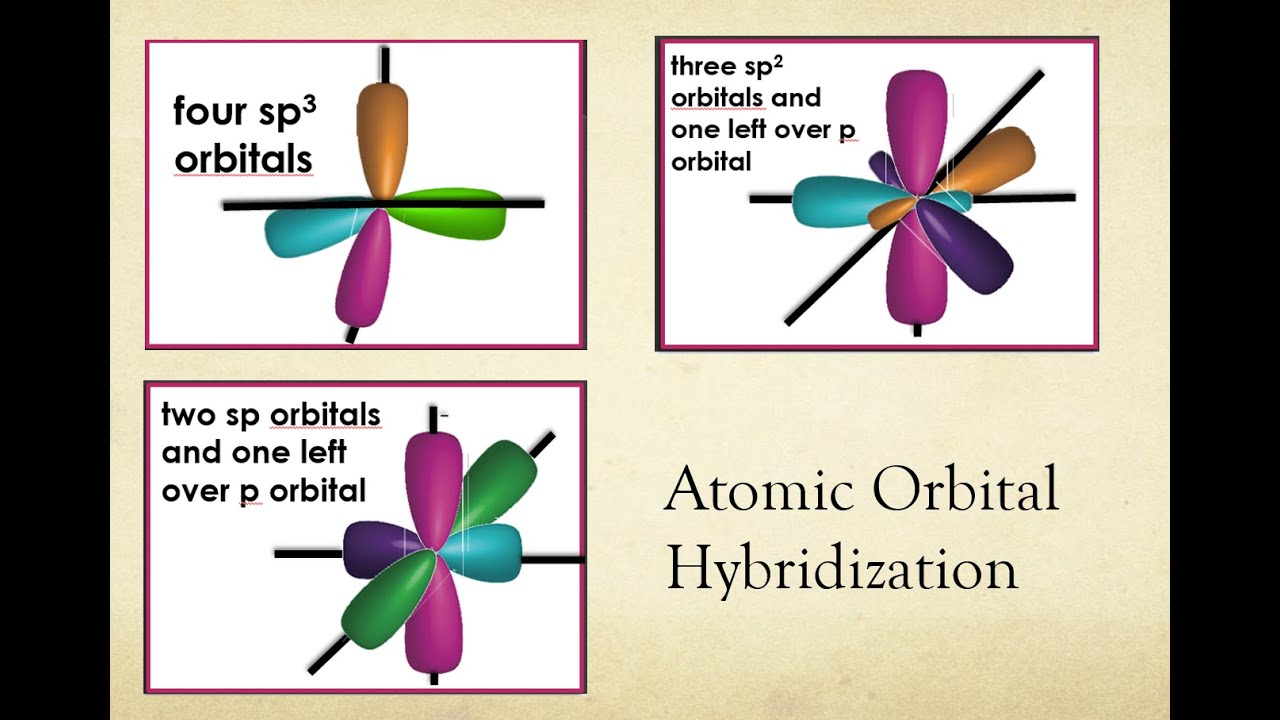
  Source: Lawrie Ryan, Cambridge A Level Chemistry Coursebook, 2014 Localised Pi Bonds Overlap of orbitals to produce sigma bonds. For example, the d orbital is introduced only in the third shell, and the f orbital only in the fourth shell of the atom. This is due to the fact that additional energy levels are able to contain more electrons and additionally, electrons in different configurations. Likewise with higher orbital shapes, they are only added in higher shells. Source: .Ī key thing to point out is that s orbitals occur in all energy shells, yet p orbitals only occur from the second shell onwards. Remember that for p and d orbitals, multiples of these orbitals come together at different axes. The different lobes that orbitals can have are c learly shown in the diagram. You don't need to concern yourself with the shape and nature of f orbitals. Orbitalīelow you will find an image of the first three types of orbitals: s, p, and d. Some orbitals have a specific shape, yet require multiple of them to come together to form an energy level. There are 4 types of orbitals as mentioned above, and each one has its own shape which is determined by the amount of lobes present in the orbital. Each electron must be in an opposing spin state due to Pauli's exclusion principle. It is key to remember that each orbital can house 2 electrons. There are 4 main types of atomic orbitals, the shapes and properties of which we will describe below. There are several types of orbitals which we will cover here. The basic definition of the orbital tells us that it is associated with defining the parameters of electrons within an atom. 
Atomic OrbitalsĪn orbital is a mathematical function of the probability of the location of an electron within the atom. Then, we will cover how they come together and how hybrid orbitals are formed. Here we will cover what orbitals are and the different types of orbitals. Finally, we'll cover some key examples of orbitals creating bonds and how hybrid orbitals provide shape to molecules.We'll look at the shape of hybrid orbitals, in the context of their molecular shape. #Hybrid atomic orbitals how to#We'll learn how to write out orbitals and the bonds they create.We'll then look at the different types of bonds created by the orbitals.We shall go over the definition of orbitals and hybrid orbitals, as well as the different types of orbitals.Ever since the shift was made from the concept of electrons being arranged in shells to orbitals, chemistry has developed immensely. Orbitals, on the other hand, are another key topic to consider regarding the arrangement of electrons within an atom. Hybridisation is a key concept within chemistry, as it underpins the dynamics of bonding as well as the shapes of molecules. In this article, we shall cover orbitals and orbital hybridisation. Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends.Molecular Structures of Acids and Bases.Ion and Atom Photoelectron Spectroscopy.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements.Transition Metal Ions in Aqueous Solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
